The U.S. Food and Drug Administration’s (FDA) vaccine advisers have unanimously voted to overhaul next season’s flu shot by including the newly dominant viral strain, subclade K, after this year’s vaccine proved far less effective than usual. The move comes as the 2025–2026 flu season winds down, with the Centers for Disease Control and Prevention (CDC) reporting that this year’s vaccine offered only 22% to 34% protection against adult outpatient visits and roughly 30% against hospitalizations—some of the lowest effectiveness rates in more than a decade.
Subclade K: The New Target
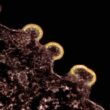
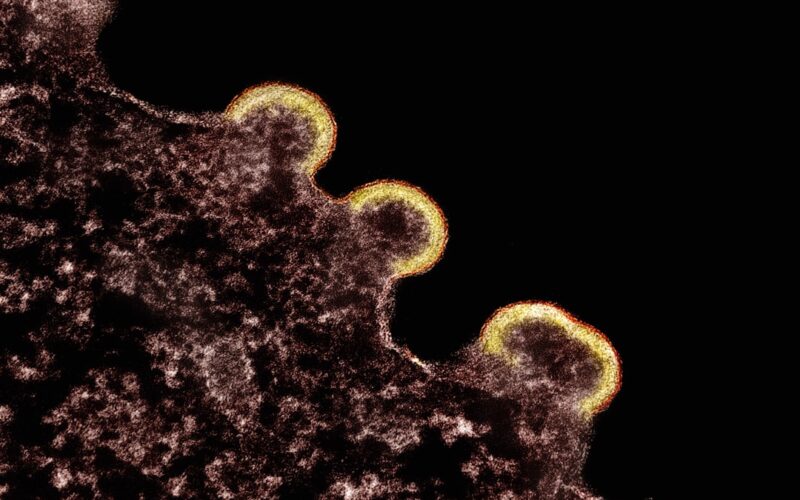
Subclade K, a mutated form of the A H3N2 flu virus, emerged as the main driver of this year’s outbreak. According to the CDC, 88% of A-positive flu specimens this season were H3N2, and a striking 93% of those were subclade K. Health officials in the U.K. and the U.S. have both noted that subclade K seems to spread more easily than its predecessors, pushing up test positivity rates and hospitalizations even as overall case numbers lag behind some previous years.
Despite the rough effectiveness numbers, the CDC stressed that vaccination still matters. Last season, flu shots prevented an estimated 5 million medical visits, 180,000 hospitalizations, and 12,000 deaths. Between October 1 and February 28, the CDC estimates the flu caused at least 26 million illnesses, 340,000 hospitalizations, and 21,000 deaths nationwide.
A Complicated Year for Vaccine Science
This year’s mismatch between circulating strains and the vaccine formula highlights the tricky business of flu vaccine development. “It’s a little bit of science, a little bit of luck,” said Arnold Monto, acting chair of the FDA’s Vaccines and Related Biological Products Advisory Committee. Strain selection is always a moving target—subclade K was first identified in June, well after last year’s vaccine formula was locked in.
The FDA panel’s March 13 recommendation, which mirrors World Health Organization guidance, means U.S. flu vaccines for 2026–2027 will cover subclade K in all major vaccine types—egg-based, cell-based, and recombinant. They’ll also better match the B strains currently circulating, aiming to head off another season of low effectiveness.
As vaccine makers gear up for next fall, the CDC continues to urge Americans to get their flu shots, citing the clear benefits even in tough years. For now, all eyes are on whether this scientific shuffle will outpace the virus’s next move.